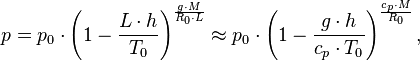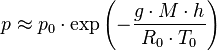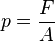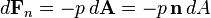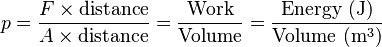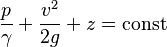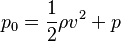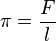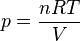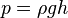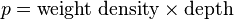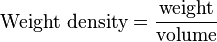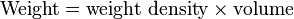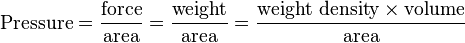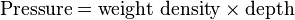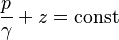Talk:Welcome to Flowpedia
"Hydraulic" redirects here. Template:Continuum mechanics Atmospheric pressure, sometimes also called barometric pressure, is the pressure exerted by the weight of air in the atmosphere of Earth (or that of another planet). In most circumstances atmospheric pressure is closely approximated by the hydrostatic pressure caused by the weight of air above the measurement point. Low-pressure areas have less atmospheric mass above their location, whereas high-pressure areas have more atmospheric mass above their location. Likewise, as elevation increases, there is less overlying atmospheric mass, so that atmospheric pressure decreases with increasing elevation. On average, a column of air Template:Convert in cross-section, measured from sea level to the top of the atmosphere, has a mass of about Template:Convert and weight of about Template:Convert. That force (across one square centimeter) is a pressure of 10.1 N/cm2 or 101,000 N/m2. A column Template:Convert in cross-section would have a weight of about Template:Cvt or about 65.4 N.
Contents
- 1 Standard atmospheric
- 2 Mean sea level pressure
- 3 Altitude variation
- 4 Local variation
- 5 Records
- 6 Measurement based on depth of water
- 7 Boiling point of water
- 8 Measurement and maps
- 9 See also
- 10 References
- 11 External links
- 12 Pressure
- 13 Definition
- 14 Types
- 15 See also
- 16 Notes
- 17 References
- 18 External links
Standard atmospheric
The standard atmosphere (symbol: atm) is a unit of pressure defined as Template:Convert, equivalent to 760 mmHg (torr), 29.92 inHg and 14.696 psi.[1]
Mean sea level pressure


The mean sea level pressure (MSLP) is the atmospheric pressure at sea level. This is the atmospheric pressure normally given in weather reports on radio, television, and newspapers or on the Internet. When barometers in the home are set to match the local weather reports, they measure pressure adjusted to sea level, not the actual local atmospheric pressure.
The altimeter setting in aviation, is an atmospheric pressure adjustment.
Average sea-level pressure is Template:Convert. In aviation weather reports (METAR), QNH is transmitted around the world in millibars or hectopascals (1 hectopascal = 1 millibar), except in the United States, Canada, and Colombia where it is reported in inches (to two decimal places) of mercury. The United States and Canada also report sea level pressure SLP, which is adjusted to sea level by a different method, in the remarks section, not in the internationally transmitted part of the code, in hectopascals or millibars.[2] However, in Canada's public weather reports, sea level pressure is instead reported in kilopascals.[3]
In the US weather code remarks, three digits are all that are transmitted; decimal points and the one or two most significant digits are omitted: Template:Convert is transmitted as 132; Template:Convert is transmitted as 000; 998.7 mbar is transmitted as 987; etc. The highest sea-level pressure on Earth occurs in Siberia, where the Siberian High often attains a sea-level pressure above Template:Convert, with record highs close to Template:Convert. The lowest measurable sea-level pressure is found at the centers of tropical cyclones and tornadoes, with a record low of Template:Convert (see Atmospheric pressure records).
Altitude variation


Pressure varies smoothly from the Earth's surface to the top of the mesosphere. Although the pressure changes with the weather, NASA has averaged the conditions for all parts of the earth year-round. As altitude increases, atmospheric pressure decreases. One can calculate the atmospheric pressure at a given altitude.[4] Temperature and humidity also affect the atmospheric pressure, and it is necessary to know these to compute an accurate figure. The graph at right was developed for a temperature of 15 °C and a relative humidity of 0%.
At low altitudes above the sea level, the pressure decreases by about 1.2 kPa for every 100 meters. For higher altitudes within the troposphere, the following equation (the barometric formula) relates atmospheric pressure p to altitude h
where the constant parameters are as described below:
| Parameter | Description | Value |
|---|---|---|
| p0 | sea level standard atmospheric pressure | 101325 Pa |
| L | temperature lapse rate, = g/cp for dry air | 0.0065 K/m |
| cp | constant pressure specific heat | ~ 1007 J/(kg•K) |
| T0 | sea level standard temperature | 288.15 K |
| g | Earth-surface gravitational acceleration | 9.80665 m/s2 |
| M | molar mass of dry air | 0.0289644 kg/mol |
| R0 | universal gas constant | 8.31447 J/(mol•K) |
Local variation
Atmospheric pressure varies widely on Earth, and these changes are important in studying weather and climate. See pressure system for the effects of air pressure variations on weather.
Atmospheric pressure shows a diurnal or semidiurnal (twice-daily) cycle caused by global atmospheric tides. This effect is strongest in tropical zones, with an amplitude of a few millibars, and almost zero in polar areas. These variations have two superimposed cycles, a circadian (24 h) cycle and semi-circadian (12 h) cycle.
Records
The highest adjusted-to-sea level barometric pressure ever recorded on Earth (above 750 meters) was Template:Convert measured in Tosontsengel, Mongolia on 19 December 2001.[5] The highest adjusted-to-sea level barometric pressure ever recorded (below 750 meters) was at Agata, Evenkiyskiy, Russia [66°53’N, 93°28’E, elevation: 261 m (856.3 ft)] on 31 December 1968 of Template:Convert.[6] The discrimination is due to the problematic assumptions (assuming a standard lapse rate) associated with reduction of sea level from high elevations.[5]
The Dead Sea, the lowest place on Earth at 425 metres (1400 feet) below sea level, has a correspondingly high typical atmospheric pressure of 1065 hPa.[7]
The lowest non-tornadic atmospheric pressure ever measured was 0.858 atm (25.69 inHg), 870 hPa, set on 12 October 1979, during Typhoon Tip in the western Pacific Ocean. The measurement was based on an instrumental observation made from a reconnaissance aircraft.[8]
Measurement based on depth of water
One atmosphere (101 kPa or 14.7 psi) is the pressure caused by the weight of a column of fresh water of approximately 10.3 m (33.8 ft). Thus, a diver 10.3 m underwater experiences a pressure of about 2 atmospheres (1 atm of air plus 1 atm of water). Conversely, 10.3 m is the maximum height to which water can be raised using suction under standard atmospheric conditions.
Low pressures such as natural gas lines are sometimes specified in inches of water, typically written as w.c. (water column) or w.g. (inches water gauge). A typical gas-using residential appliance in the US is rated for a maximum of 14 w.c., which is approximately 35 hPa. Similar metric units with a wide variety of names and notation based on millimetres, centimetres or metres are now less commonly used.
Boiling point of water
Pure water boils at Template:Convert at earth's standard atmospheric pressure. The boiling point is the temperature at which the vapor pressure is equal to the atmospheric pressure around the water.[9] Because of this, the boiling point of water is lower at lower pressure and higher at higher pressure. Cooking at high elevations, therefore, requires adjustments to recipes.[10] A rough approximation of elevation can be obtained by measuring the temperature at which water boils; in the mid-19th century, this method was used by explorers.[11]
Measurement and maps
An important application of the knowledge that atmospheric pressure varies directly with altitude was in determining the height of hills and mountains thanks to the availability of reliable pressure measurement devices. While in 1774 Maskelyne was confirming Newton's theory of gravitation at and on Schiehallion in Scotland (using plumb bob deviation to show the effect of "gravity") and accurately measure elevation, William Roy using barometric pressure was able to confirm his height determinations, the agreement being to within one meter (3.28 feet). This was then a useful tool for survey work and map making and long has continued to be useful. It was part of the "application of science" which gave practical people the insight that applied science could easily and relatively cheaply be "useful".[12]
See also
Template:Portal Template:Div col
- Atmosphere (unit)
- Barometric formula
- Barotrauma – physical damage to body tissues caused by a difference in pressure between an air space inside or beside the body and the surrounding gas or liquid.
- Cabin pressurization
- Effects of high altitude on humans
- High-pressure area
- International Standard Atmosphere - a tabulation of typical variation of principal thermodynamic variables of the atmosphere (pressure, density, temperature, etc.) with altitude, at middle latitudes.
- Low-pressure area
- NRLMSISE-00
- Plenum chamber
- Pressure
- Subtropical high belts
References
External links
- How Atmospheric Pressure Affects Objects (Audio slideshow from the National High Magnetic Field Laboratory)
- 1976 Standard Atmosphere from NASA
- Source code and equations for the 1976 Standard Atmosphere
- A mathematical model of the 1976 U.S. Standard Atmosphere
- Calculator using multiple units and properties for the 1976 Standard Atmosphere
- Calculator giving standard air pressure at a specified altitude, or altitude at which a pressure would be standard
- Some of the effects of air pressure
- Atmospheric calculator and Geometric to Pressure altitude converter
Experiments
- Movies on atmospheric pressure experiments from Georgia State University's HyperPhysics website – requires QuickTime
- Test showing a can being crushed after boiling water inside it, then moving it into a tub of ice cold water.
Template:Meteorological variables Template:Diving medicine, physiology and physics
Pressure
Template:Infobox physical quantity
Pressure (symbol: p or P) is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled gage pressure)[lower-alpha 1] is the pressure relative to the ambient pressure.
Various units are used to express pressure. Some of these derive from a unit of force divided by a unit of area; the SI unit of pressure, the pascal (Pa), for example, is one newton per square metre; similarly, the pound-force per square inch (psi) is the traditional unit of pressure in the imperial and US customary systems. Pressure may also be expressed in terms of standard atmospheric pressure; the atmosphere (atm) is equal to this pressure and the torr is defined as Template:Frac of this. Manometric units such as the centimetre of water, millimetre of mercury, and inch of mercury are used to express pressures in terms of the height of column of a particular fluid in a manometer.
Definition
Pressure is the amount of force acting per unit area. The symbol for it is p or P.[13] The IUPAC recommendation for pressure is a lower-case p.[14] However, upper-case P is widely used. The usage of P vs p depends upon the field in which one is working, on the nearby presence of other symbols for quantities such as power and momentum, and on writing style.
Formula
Template:Conjugate variables (thermodynamics)
Mathematically:
where:
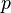 is the pressure,
is the pressure,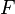 is the normal force,
is the normal force,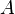 is the area of the surface on contact.
is the area of the surface on contact.
Pressure is a scalar quantity. It relates the vector surface element (a vector normal to the surface) with the normal force acting on it. The pressure is the scalar proportionality constant that relates the two normal vectors:
The minus sign comes from the fact that the force is considered towards the surface element, while the normal vector points outward. The equation has meaning in that, for any surface S in contact with the fluid, the total force exerted by the fluid on that surface is the surface integral over S of the right-hand side of the above equation.
It is incorrect (although rather usual) to say "the pressure is directed in such or such direction". The pressure, as a scalar, has no direction. The force given by the previous relationship to the quantity has a direction, but the pressure does not. If we change the orientation of the surface element, the direction of the normal force changes accordingly, but the pressure remains the same.
Pressure is distributed to solid boundaries or across arbitrary sections of fluid normal to these boundaries or sections at every point. It is a fundamental parameter in thermodynamics, and it is conjugate to volume.
Units
The SI unit for pressure is the pascal (Pa), equal to one newton per square metre (N/m2 or kg·m−1·s−2). This name for the unit was added in 1971;[15] before that, pressure in SI was expressed simply in newtons per square metre.
Other units of pressure, such as pounds per square inch and bar, are also in common use. The CGS unit of pressure is the barye (Ba), equal to 1 dyn·cm−2 or 0.1 Pa. Pressure is sometimes expressed in grams-force or kilograms-force per square centimetre (g/cm2 or kg/cm2) and the like without properly identifying the force units. But using the names kilogram, gram, kilogram-force, or gram-force (or their symbols) as units of force is expressly forbidden in SI. The technical atmosphere (symbol: at) is 1 kgf/cm2 (98.0665 kPa or 14.223 psi).
Since a system under pressure has potential to perform work on its surroundings, pressure is a measure of potential energy stored per unit volume. It is therefore related to energy density and may be expressed in units such as joules per cubic metre (J/m3, which is equal to Pa). Mathematically:
Some meteorologists prefer the hectopascal (hPa) for atmospheric air pressure, which is equivalent to the older unit millibar (mbar). Similar pressures are given in kilopascals (kPa) in most other fields, where the hecto- prefix is rarely used. The inch of mercury is still used in the United States. Oceanographers usually measure underwater pressure in decibars (dbar) because pressure in the ocean increases by approximately one decibar per metre depth.
The standard atmosphere (atm) is an established constant. It is approximately equal to typical air pressure at earth mean sea level and is defined as Template:Val.
Because pressure is commonly measured by its ability to displace a column of liquid in a manometer, pressures are often expressed as a depth of a particular fluid (e.g., centimetres of water, millimetres of mercury or inches of mercury). The most common choices are mercury (Hg) and water; water is nontoxic and readily available, while mercury's high density allows a shorter column (and so a smaller manometer) to be used to measure a given pressure. The pressure exerted by a column of liquid of height h and density ρ is given by the hydrostatic pressure equation Template:Nowrap, where g is the gravitational acceleration. Fluid density and local gravity can vary from one reading to another depending on local factors, so the height of a fluid column does not define pressure precisely. When millimetres of mercury or inches of mercury are quoted today, these units are not based on a physical column of mercury; rather, they have been given precise definitions that can be expressed in terms of SI units.Template:Citation needed One millimetre of mercury is approximately equal to one torr. The water-based units still depend on the density of water, a measured, rather than defined, quantity. These manometric units are still encountered in many fields. Blood pressure is measured in millimetres of mercury in most of the world, and lung pressures in centimetres of water are still common.
Underwater divers use the metre sea water (msw or MSW) and foot sea water (fsw or FSW) units of pressure, and these are the standard units for pressure gauges used to measure pressure exposure in diving chambers and personal decompression computers. A msw is defined as 0.1 bar (0.1 bar [= 100000 Pa] = 10000 Pa), and is not the same as a linear metre of depth, and 33.066 fsw = 1 atm.[16] (1 atm [= 101325 Pa] / 33.066 = 3064.326 Pa) Note that the pressure conversion from msw to fsw is different from the length conversion: 10 msw = 32.6336 fsw, while 10 m = 32.8083 ft.[17]
Gauge pressure is often given in units with 'g' appended, e.g. 'kPag', 'barg' or 'psig', and units for measurements of absolute pressure are sometimes given a suffix of 'a', to avoid confusion, for example 'kPaa', 'psia'. However, the US National Institute of Standards and Technology recommends that, to avoid confusion, any modifiers be instead applied to the quantity being measured rather than the unit of measure[18] For example, Template:Nowrap rather than Template:Nowrap.
Differential pressure is expressed in units with 'd' appended; this type of measurement is useful when considering sealing performance or whether a valve will open or close.
Presently or formerly popular pressure units include the following:
- atmosphere (atm)
- manometric units:
- centimetre, inch, millimetre (torr) and micrometre (mTorr, micron) of mercury
- Template:AnchorHeight of equivalent column of water, including millimetre (mm Template:Chem), centimetre (cm Template:Chem), metre, inch, and foot of water
- imperial and customary units:
- kip, short ton-force, long ton-force, pound-force, ounce-force, and poundal per square inch
- short ton-force and long ton-force per square inch
- fsw (feet sea water) used in underwater diving, particularly in connection with diving pressure exposure and decompression
- non-SI metric units:
- bar, decibar, millibar
- msw (metres sea water), used in underwater diving, particularly in connection with diving pressure exposure and decompression
- kilogram-force, or kilopond, per square centimetre (technical atmosphere)
- gram-force and tonne-force (metric ton-force) per square centimetre
- barye (dyne per square centimetre)
- kilogram-force and tonne-force per square metre
- sthene per square metre (pieze)
- bar, decibar, millibar
Examples
As an example of varying pressures, a finger can be pressed against a wall without making any lasting impression; however, the same finger pushing a thumbtack can easily damage the wall. Although the force applied to the surface is the same, the thumbtack applies more pressure because the point concentrates that force into a smaller area. Pressure is transmitted to solid boundaries or across arbitrary sections of fluid normal to these boundaries or sections at every point. Unlike stress, pressure is defined as a scalar quantity. The negative gradient of pressure is called the force density.
Another example is of a common knife. If we try to cut a fruit with the flat side it obviously will not cut. But if we take the thin side, it will cut smoothly. The reason is that the flat side has a greater surface area (less pressure) and so it does not cut the fruit. When we take the thin side, the surface area is reduced and so it cuts the fruit easily and quickly. This is one example of a practical application of pressure.
For gases, pressure is sometimes measured not as an absolute pressure, but relative to atmospheric pressure; such measurements are called gauge pressure. An example of this is the air pressure in an automobile tire, which might be said to be "220 kPa (32 psi)", but is actually 220 kPa (32 psi) above atmospheric pressure. Since atmospheric pressure at sea level is about 100 kPa (14.7 psi), the absolute pressure in the tire is therefore about 320 kPa (46.7 psi). In technical work, this is written "a gauge pressure of 220 kPa (32 psi)". Where space is limited, such as on pressure gauges, name plates, graph labels, and table headings, the use of a modifier in parentheses, such as "kPa (gauge)" or "kPa (absolute)", is permitted. In non-SI technical work, a gauge pressure of 32 psi is sometimes written as "32 psig" and an absolute pressure as "32 psia", though the other methods explained above that avoid attaching characters to the unit of pressure are preferred.[19]
Gauge pressure is the relevant measure of pressure wherever one is interested in the stress on storage vessels and the plumbing components of fluidics systems. However, whenever equation-of-state properties, such as densities or changes in densities, must be calculated, pressures must be expressed in terms of their absolute values. For instance, if the atmospheric pressure is 100 kPa, a gas (such as helium) at 200 kPa (gauge) (300 kPa [absolute]) is 50% denser than the same gas at 100 kPa (gauge) (200 kPa [absolute]). Focusing on gauge values, one might erroneously conclude the first sample had twice the density of the second one.
Scalar nature
In a static gas, the gas as a whole does not appear to move. The individual molecules of the gas, however, are in constant random motion. Because we are dealing with an extremely large number of molecules and because the motion of the individual molecules is random in every direction, we do not detect any motion. If we enclose the gas within a container, we detect a pressure in the gas from the molecules colliding with the walls of our container. We can put the walls of our container anywhere inside the gas, and the force per unit area (the pressure) is the same. We can shrink the size of our "container" down to a very small point (becoming less true as we approach the atomic scale), and the pressure will still have a single value at that point. Therefore, pressure is a scalar quantity, not a vector quantity. It has magnitude but no direction sense associated with it. Pressure acts in all directions at a point inside a gas. At the surface of a gas, the pressure force acts perpendicular (at right angle) to the surface.
A closely related quantity is the stress tensor σ, which relates the vector force 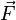 to the
vector area
to the
vector area 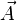 via the linear relation
via the linear relation 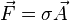 .
.
This tensor may be expressed as the sum of the viscous stress tensor minus the hydrostatic pressure. The negative of the stress tensor is sometimes called the pressure tensor, but in the following, the term "pressure" will refer only to the scalar pressure.
According to the theory of general relativity, pressure increases the strength of a gravitational field (see stress–energy tensor) and so adds to the mass-energy cause of gravity. This effect is unnoticeable at everyday pressures but is significant in neutron stars, although it has not been experimentally tested.[20]
Types
Fluid pressure
Fluid pressure is the pressure at some point within a fluid, such as water or air (for more information specifically about liquid pressure, see section below).
Fluid pressure occurs in one of two situations:
- an open condition, called "open channel flow", e.g. the ocean, a swimming pool, or the atmosphere.
- a closed condition, called "closed conduit", e.g. a water line or gas line.
Pressure in open conditions usually can be approximated as the pressure in "static" or non-moving conditions (even in the ocean where there are waves and currents), because the motions create only negligible changes in the pressure. Such conditions conform with principles of fluid statics. The pressure at any given point of a non-moving (static) fluid is called the hydrostatic pressure.
Closed bodies of fluid are either "static", when the fluid is not moving, or "dynamic", when the fluid can move as in either a pipe or by compressing an air gap in a closed container. The pressure in closed conditions conforms with the principles of fluid dynamics.
The concepts of fluid pressure are predominantly attributed to the discoveries of Blaise Pascal and Daniel Bernoulli. Bernoulli's equation can be used in almost any situation to determine the pressure at any point in a fluid. The equation makes some assumptions about the fluid, such as the fluid being ideal[21] and incompressible.[21] An ideal fluid is a fluid in which there is no friction, it is inviscid [21] (zero viscosity).[21] The equation for all points of a system filled with a constant-density fluid is
where:
- p = pressure of the fluid
- γ = ρg = density·acceleration of gravity = specific weight of the fluid.[21]
- v = velocity of the fluid
- g = acceleration of gravity
- z = elevation
 = pressure head
= pressure head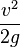 = velocity head
= velocity head
Applications
Explosion or deflagration pressures
Explosion or deflagration pressures are the result of the ignition of explosive gases, mists, dust/air suspensions, in unconfined and confined spaces.
Negative pressures

While pressures are, in general, positive, there are several situations in which negative pressures may be encountered:
- When dealing in relative (gauge) pressures. For instance, an absolute pressure of 80 kPa may be described as a gauge pressure of −21 kPa (i.e., 21 kPa below an atmospheric pressure of 101 kPa).
- When attractive intermolecular forces (e.g., van der Waals forces or hydrogen bonds) between the particles of a fluid exceed repulsive forces due to thermal motion. These forces explain ascent of sap in tall plants. An apparent negative pressure must act on water molecules at the top of any tree taller than 10 m, which is the pressure head of water that balances the atmospheric pressure. Intermolecular forces maintain cohesion of columns of sap that run continuously in xylem from the roots to the top leaves.[23]
- The Casimir effect can create a small attractive force due to interactions with vacuum energy; this force is sometimes termed "vacuum pressure" (not to be confused with the negative gauge pressure of a vacuum).
- For non-isotropic stresses in rigid bodies, depending on how the orientation of a surface is chosen, the same distribution of forces may have a component of positive pressure along one surface normal, with a component of negative pressure acting along another surface normal.
- The stresses in an electromagnetic field are generally non-isotropic, with the pressure normal to one surface element (the normal stress) being negative, and positive for surface elements perpendicular to this.
- In the cosmological constant.
Stagnation pressure
Stagnation pressure is the pressure a fluid exerts when it is forced to stop moving. Consequently, although a fluid moving at higher speed will have a lower static pressure, it may have a higher stagnation pressure when forced to a standstill. Static pressure and stagnation pressure are related by:
where
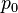 is the stagnation pressure
is the stagnation pressure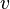 is the flow velocity
is the flow velocity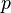 is the static pressure.
is the static pressure.
The pressure of a moving fluid can be measured using a Pitot tube, or one of its variations such as a Kiel probe or Cobra probe, connected to a manometer. Depending on where the inlet holes are located on the probe, it can measure static pressures or stagnation pressures.
Surface pressure and surface tension
There is a two-dimensional analog of pressure – the lateral force per unit length applied on a line perpendicular to the force.
Surface pressure is denoted by π and shares many similar properties with three-dimensional pressure. Properties of surface chemicals can be investigated by measuring pressure/area isotherms, as the two-dimensional analog of Boyle's law, Template:Nowrap, at constant temperature.
Surface tension is another example of surface pressure, but with a reversed sign, because "tension" is the opposite to "pressure".
Pressure of an ideal gas
Template:Main article In an ideal gas, molecules have no volume and do not interact. According to the ideal gas law, pressure varies linearly with temperature and quantity, and inversely with volume.
where:
- p is the absolute pressure of the gas
- n is the amount of substance
- T is the absolute temperature
- V is the volume
- R is the ideal gas constant.
Real gases exhibit a more complex dependence on the variables of state.[24]
Vapor pressure
Template:Main article Vapor pressure is the pressure of a vapor in thermodynamic equilibrium with its condensed phases in a closed system. All liquids and solids have a tendency to evaporate into a gaseous form, and all gases have a tendency to condense back to their liquid or solid form.
The atmospheric pressure boiling point of a liquid (also known as the normal boiling point) is the temperature at which the vapor pressure equals the ambient atmospheric pressure. With any incremental increase in that temperature, the vapor pressure becomes sufficient to overcome atmospheric pressure and lift the liquid to form vapor bubbles inside the bulk of the substance. Bubble formation deeper in the liquid requires a higher pressure, and therefore higher temperature, because the fluid pressure increases above the atmospheric pressure as the depth increases.
The vapor pressure that a single component in a mixture contributes to the total pressure in the system is called partial vapor pressure.
Liquid pressure
Template:See also Template:Continuum mechanics When a person swims under the water, water pressure is felt acting on the person's eardrums. The deeper that person swims, the greater the pressure. The pressure felt is due to the weight of the water above the person. As someone swims deeper, there is more water above the person and therefore greater pressure. The pressure a liquid exerts depends on its depth.
Liquid pressure also depends on the density of the liquid. If someone was submerged in a liquid more dense than water, the pressure would be correspondingly greater. The pressure due to a liquid in liquid columns of constant density or at a depth within a substance is represented by the following formula:
where:
- p is liquid pressure
- g is gravity at the surface of overlaying material
- ρ is density of liquid
- h is height of liquid column or depth within a substance.
Another way of saying this same formula is the following:
| Derivation of this equation |
|---|
| This is derived from the definitions of pressure and weight density. Consider an area at the bottom of a vessel of liquid. The weight of the column of liquid directly above this area produces pressure. From the definition:
we can express this weight of liquid as where the volume of the column is simply the area multiplied by the depth. Then we have With the "area" in the numerator and the "area" in the denominator canceling each other out, we are left with: Written with symbols, this is our original equation: |
The pressure a liquid exerts against the sides and bottom of a container depends on the density and the depth of the liquid. If atmospheric pressure is neglected, liquid pressure against the bottom is twice as great at twice the depth; at three times the depth, the liquid pressure is threefold; etc. Or, if the liquid is two or three times as dense, the liquid pressure is correspondingly two or three times as great for any given depth. Liquids are practically incompressible – that is, their volume can hardly be changed by pressure (water volume decreases by only 50 millionths of its original volume for each atmospheric increase in pressure). Thus, except for small changes produced by temperature, the density of a particular liquid is practically the same at all depths.
Atmospheric pressure pressing on the surface of a liquid must be taken into account when trying to discover the total pressure acting on a liquid. The total pressure of a liquid, then, is ρgh plus the pressure of the atmosphere. When this distinction is important, the term total pressure is used. Otherwise, discussions of liquid pressure refer to pressure without regard to the normally ever-present atmospheric pressure.
It is important to recognize that the pressure does not depend on the amount of liquid present. Volume is not the important factor – depth is. The average water pressure acting against a dam depends on the average depth of the water and not on the volume of water held back. For example, a wide but shallow lake with a depth of Template:Convert exerts only half the average pressure that a small Template:Convert deep pond does (note that the total force applied to the longer dam will be greater, due to the greater total surface area for the pressure to act upon, but for a given 5-foot section of each dam, the 10 ft deep water will apply half the force of 20 ft deep water). A person will feel the same pressure whether his/her head is dunked a metre beneath the surface of the water in a small pool or to the same depth in the middle of a large lake. If four vases contain different amounts of water but are all filled to equal depths, then a fish with its head dunked a few centimetres under the surface will be acted on by water pressure that is the same in any of the vases. If the fish swims a few centimetres deeper, the pressure on the fish will increase with depth and be the same no matter which vase the fish is in. If the fish swims to the bottom, the pressure will be greater, but it makes no difference what vase it is in. All vases are filled to equal depths, so the water pressure is the same at the bottom of each vase, regardless of its shape or volume. If water pressure at the bottom of a vase were greater than water pressure at the bottom of a neighboring vase, the greater pressure would force water sideways and then up the narrower vase to a higher level until the pressures at the bottom were equalized. Pressure is depth dependent, not volume dependent, so there is a reason that water seeks its own level.
Restating this as energy equation, the energy per unit volume in an ideal, incompressible liquid is constant throughout its vessel. At the surface, gravitational potential energy is large but liquid pressure energy is low. At the bottom of the vessel, all the gravitational potential energy is converted to pressure energy. The sum of pressure energy and gravitational potential energy per unit volume is constant throughout the volume of the fluid and the two energy components change linearly with the depth.[25] Mathematically, it is described by Bernoulli's equation where velocity head is zero and comparisons per unit volume in the vessel are:
Terms have the same meaning as in section Fluid pressure.
Direction of liquid pressure
An experimentally determined fact about liquid pressure is that it is exerted equally in all directions.[26] If someone is submerged in water, no matter which way that person tilts his/her head, the person will feel the same amount of water pressure on his/her ears. Because a liquid can flow, this pressure isn't only downward. Pressure is seen acting sideways when water spurts sideways from a leak in the side of an upright can. Pressure also acts upward, as demonstrated when someone tries to push a beach ball beneath the surface of the water. The bottom of a boat is pushed upward by water pressure (buoyancy).
When a liquid presses against a surface, there is a net force that is perpendicular to the surface. Although pressure doesn't have a specific direction, force does. A submerged triangular block has water forced against each point from many directions, but components of the force that are not perpendicular to the surface cancel each other out, leaving only a net perpendicular point.[26] This is why water spurting from a hole in a bucket initially exits the bucket in a direction at right angles to the surface of the bucket in which the hole is located. Then it curves downward due to gravity. If there are three holes in a bucket (top, bottom, and middle), then the force vectors perpendicular to the inner container surface will increase with increasing depth – that is, a greater pressure at the bottom makes it so that the bottom hole will shoot water out the farthest. The force exerted by a fluid on a smooth surface is always at right angles to the surface. The speed of liquid out of the hole is 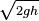 , where h is the depth below the free surface.[26] Interestingly, this is the same speed the water (or anything else) would have if freely falling the same vertical distance h.
, where h is the depth below the free surface.[26] Interestingly, this is the same speed the water (or anything else) would have if freely falling the same vertical distance h.
Kinematic pressure
is the kinematic pressure, where 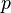 is the pressure and
is the pressure and 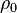 constant mass density. The SI unit of P is m2/s2. Kinematic pressure is used in the same manner as kinematic viscosity
constant mass density. The SI unit of P is m2/s2. Kinematic pressure is used in the same manner as kinematic viscosity 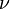 in order to compute Navier–Stokes equation without explicitly showing the density
in order to compute Navier–Stokes equation without explicitly showing the density 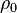 .
.
- Navier–Stokes equation with kinematic quantities
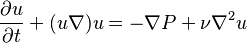
See also
Notes
References
External links
Template:Diving medicine, physiology and physics
Template:Authority control- ↑ International Civil Aviation Organization. Manual of the ICAO Standard Atmosphere, Doc 7488-CD, Third Edition, 1993. ISBN 92-9194-004-6.
- ↑ Sample METAR of CYVR Nav Canada
- ↑ Template:Citation
- ↑ A quick derivation relating altitude to air pressure by Portland State Aerospace Society, 2004, accessed 05032011
- ↑ 5.0 5.1 Template:Citation
- ↑ Template:Citation
- ↑ Template:Cite journal
- ↑ Template:Cite web
- ↑ Template:Citation
- ↑ Template:Citation
- ↑ Template:Cite journal
- ↑ Hewitt, Rachel, Map of a Nation – a Biography of the Ordnance Survey ISBN 1-84708-098-7
- ↑ Template:Cite book
- ↑ Template:Cite book
- ↑ Template:Cite web
- ↑ Template:Cite book
- ↑ Template:Cite web
- ↑ Template:Cite web
- ↑ NIST, Rules and Style Conventions for Expressing Values of Quantities, Sect. 7.4.
- ↑ Template:Cite web
- ↑ 21.0 21.1 21.2 21.3 21.4 Template:Cite book
- ↑ Template:Cite book
- ↑ Template:Cite web
- ↑ P. Atkins, J. de Paula Elements of Physical Chemistry, 4th Ed, W.H. Freeman, 2006. ISBN 0-7167-7329-5.
- ↑ Streeter, V.L., Fluid Mechanics, Example 3.5, McGraw–Hill Inc. (1966), New York.
- ↑ 26.0 26.1 26.2 Hewitt 251 (2006)
Cite error: <ref> tags exist for a group named "lower-alpha", but no corresponding <references group="lower-alpha"/> tag was found, or a closing </ref> is missing